Scientists prevent heart failure in mice 25th September 2012 Cardiac stress — for example, a heart attack or high blood pressure — often leads to pathological heart growth and subsequently to heart failure. Two tiny RNA molecules play a key role in this detrimental development in mice, as researchers at the Hannover Medical School and the Göttingen Max Planck Institute for Biophysical Chemistry have now discovered.
When they inhibited one of these two specific molecules, they were able to protect the rodents against pathological heart growth and failure. With these findings, the scientists hope to develop therapeutic approaches that could one day protect humans against heart failure. Respiratory distress, fatigue, and attenuated performance are symptoms that can accompany heart failure. A reason for this can be an enlarged heart, also known as cardiac hypertrophy. This may develop when the heart is subjected to permanent stress. For example, due to persistent high blood pressure or a valvular heart defect. In order to boost the pumping performance, heart muscle cells enlarge — a condition that frequently results in heart failure if not treated. A research team at the Göttingen Max Planck Institute for Biophysical Chemistry and the Hannover Medical School discovered that two small RNA molecules play a key role in the growth of heart muscle cells: the microRNAs miR-212 and miR-132. The scientists had observed that these microRNAs are more prevalent in the cardiac muscle cells of mice suffering from cardiac hypertrophy. To determine the role that the two microRNAs play, the scientists bred genetically modified mice that had an abnormally large number of these molecules in their heart muscle cells.
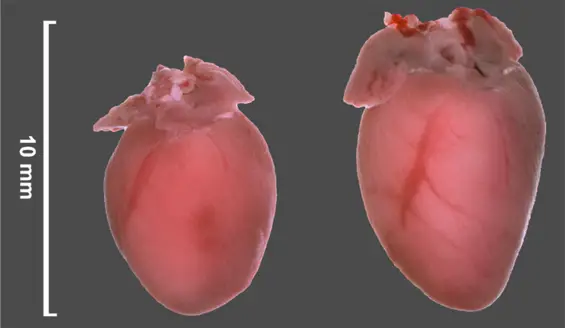
"These rodents developed cardiac hypertrophy and lived for only three to six months, whereas their healthy conspecifics had a normal healthy life-span of several years," explained Dr. Kamal Chowdhury, researcher in the Department of Molecular Cell Biology at the Max Planck Institute for Biophysical Chemistry. "For comparison, we also selectively switched off these microRNAs in other mice. These animals had a slightly smaller heart than their healthy conspecifics, but did not differ from them in behavior or life-span," continued the biologist. The crucial point is when the scientists subjected the hearts of these mice to stress by narrowing the aorta, the mice did not develop cardiac hypertrophy — in contrast to normal mice. The scientists were also able to protect normal mice against the disease. When they gave them a substance that selectively inhibits microRNA-132, no pathological cardiac growth occurred — even when the hearts of these mice were subjected to stress. "Thus, for the first time ever, we have found a molecular approach for treating pathological cardiac growth and heart failure in mice," said cardiologist Professor Dr. Thomas Thum at the Hannover Medical School. With these findings, the researchers hope to develop therapeutic approaches in the future that can also protect humans against heart failure. "Such microRNA inhibitors — alone, or in combination with conventional treatments — could represent a promising new therapeutic approach," said Thum.
Comments »
|