14th March 2019 A step closer to resurrecting the woolly mammoth A team of Japanese and Russian scientists report that cell nuclei from woolly mammoth remains showed biological activity when transplanted into mouse cells.
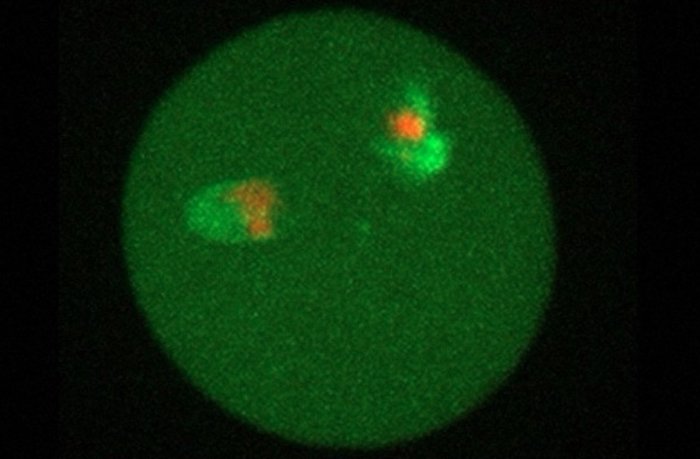
A collaborative research group led by Kindai University, Japan, has succeeded in observing biological activity in cell nuclei collected from the 28,000-year-old remains of a mammoth named "Yuka", found in Siberian permafrost. The mammoth cell nuclei were transplanted into mouse oocytes; a method of nuclear transfer that is used for producing cloned animals. After nuclear transfer, mouse proteins were loaded onto the mammoth cell nucleus and a part of the mammoth cell nucleus started to form a new nucleus-like structure. Such events are observed when animals are cloned using this method, and therefore this confirms that the ancient specimen still possesses at least partially active nuclei. Cell nuclei contain DNA, which forms the blueprint of life and determines the structure of proteins in an organism. However, until now, it has not been known whether cell nuclei extracted from fossils can retain biological functionality. In this study, researchers obtained bone marrow and muscle tissue from the frozen body of "Yuka" and then performed detailed analyses to find the least-damaged nucleus-like structures. "First, we compared the genetic information of Yuka with African elephants and confirmed the characteristic single base substitution and amino acid substitution possessed by mammoths," the team writes. "Next, mass spectroscopy analysis of the protein preservation state showed that the muscle tissue of Yuka was preserved in a relatively good state and the components of the cell nucleus were present."
A total of 88 nuclei structures were collected from 274 milligrams of mammoth tissue. These were injected into egg cells using a micromanipulator and the reaction was then observed using a disc type confocal laser microscope. Some of the mammoth cell nuclei began to take up mouse cell nuclear proteins in the mouse oocytes, and cells shaped as those do before division normally occurs were observed. Furthermore, part of the mammoth nuclei started to form a partial nucleus-like structure, and this newly-formed structure was eventually incorporated into the cell nucleus of the mouse oocyte. "In the reconstructed oocytes, the mammoth nuclei showed the spindle assembly, histone incorporation and partial nuclear formation; however, full activation of nuclei for cleavage was not confirmed," the team says. "This study revealed for the first time in the world that the cell nuclei of mammoth have maintained its biological activity for 28,000 years inside permafrost, and it has also been revealed that the damage to genomic DNA can be partially repaired using the ability of the mouse oocyte. As a result, DNA replication, gene transcription, translation and embryonic development of ancient creatures may be possible if the obtained cell nuclei have mildly damaged genomic DNA and a good preservation state.
"In the future, by utilising Kindai University's 'Bioassay Platform' – using bioinformatics science and nuclear transfer that integrates state-of-the-art knowledge and technology – the reproduction of cellular-level phenomena of extinct creatures may be possible," the researchers conclude. Their study is published this week in the peer-reviewed journal, Scientific Reports. This marks a "significant step toward bringing mammoths back from the dead," said Kei Miyamoto, one of the lead authors. "Once we obtain cell nuclei that are kept in better condition, we can expect to advance the research to the stage of cell division."
Comments »
If you enjoyed this article, please consider sharing it:
|