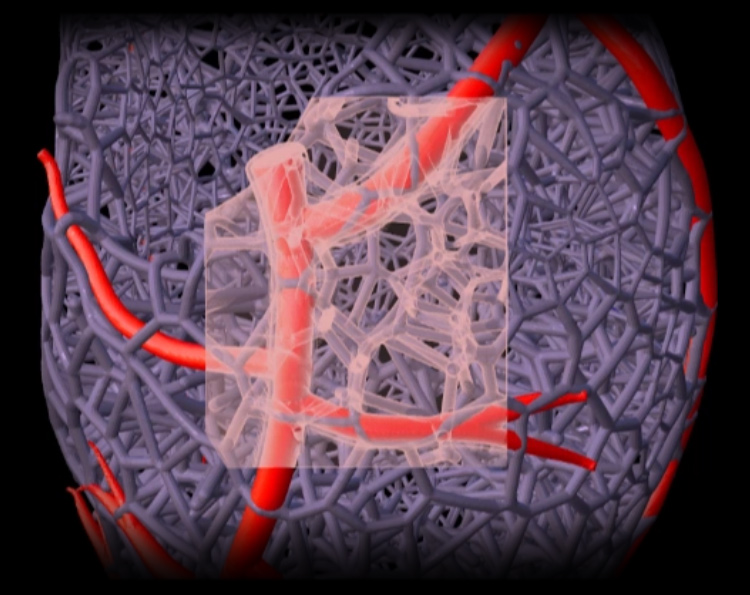
3rd August 2019 A step closer to 3D printed human hearts Researchers from Carnegie Mellon University have published a paper in Science that details a new technique for 3D bioprinting tissue scaffolds made out of collagen, the major structural protein in the human body. This first-of-its-kind method brings the field of tissue engineering one step closer to being able to 3D print a full-sized, adult human heart. The technique, known as Freeform Reversible Embedding of Suspended Hydrogels (FRESH), has allowed the researchers to overcome many challenges associated with existing 3D bioprinting methods, and to achieve unprecedented resolution and fidelity using soft and living materials. Each of the organs in the human body, such as the heart, is built from specialised cells that are held together by a biological scaffold called the extracellular matrix (ECM). This network of ECM proteins provides the structure and biochemical signals that cells need to carry out their normal function. Until now, however, it has not been possible to rebuild this complex ECM architecture using traditional biofabrication methods.
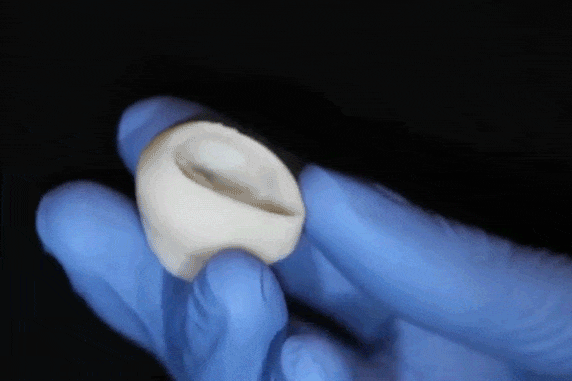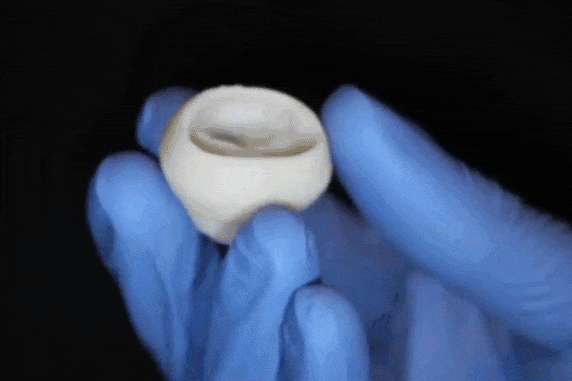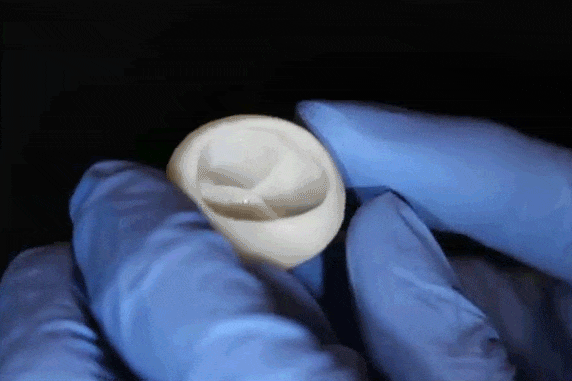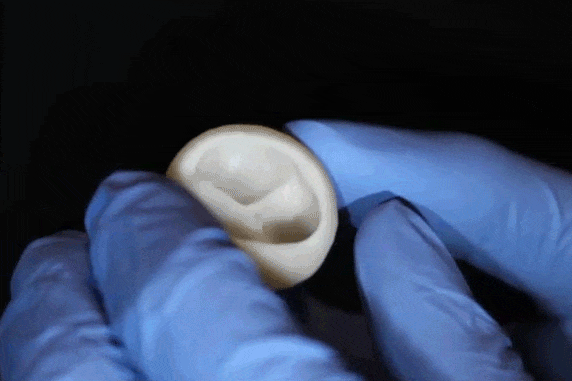
"What we've shown is that we can print pieces of the heart out of cells and collagen into parts that truly function – like a heart valve, or small beating ventricle," says Professor Adam Feinberg, of the Departments of Biomedical Engineering and Materials Science and Engineering, whose laboratory conducted this research. "Using MRI data of a human heart, we were able to accurately reproduce patient-specific anatomical structure and 3D bioprint collagen and human heart cells." Over 4,000 patients in the USA are waiting for a heart transplant, while millions more worldwide need hearts but are ineligible for the waitlist. The demand for replacement organs is immense, and new approaches are needed to engineer artificial organs that are capable of repairing, supplementing or replacing long-term organ function. Feinberg is working to solve these challenges with a new generation of bioengineered organs that more closely replicate natural organ structures. "Collagen is an extremely desirable biomaterial to 3D print with, because it makes up literally every single tissue in your body," says Andrew Hudson, a PhD student in Feinberg's lab. "What makes it so hard to 3D print, however, is that it starts out as a fluid – so if you try to print this in air, it just forms a puddle on your build platform. So we've developed a technique that prevents it from deforming." The FRESH method developed in Feinberg's lab allows collagen to be deposited layer-by-layer within a support bath of gel, giving the collagen a chance to solidify in place before it is removed. With FRESH, the support gel can be easily melted away by heating it from room temperature to body temperature after the print has been completed. This way, researchers can remove the support gel without damaging the printed structure made of collagen or cells.
This method is truly exciting for the field of 3D bioprinting, because it allows collagen scaffolds to be printed at the large scale of human organs. And it is not limited to collagen, as a wide range of other soft gels – including fibrin, alginate, and hyaluronic acid – can be printed using the FRESH technique, providing a robust and adaptable tissue engineering platform. Importantly, Feinberg's team also made their designs open-source, so that nearly anyone, from medical labs to high school science classes can build and have access to low-cost, high-performance 3D bioprinters. Looking forward, FRESH has applications in many aspects of regenerative medicine, from wound repair to organ bioengineering, but it is just one piece of a growing biofabrication field, as Feinberg explains: "Really what we're talking about is the convergence of technologies. Not just what my lab does in bioprinting, but also from other labs and small companies in the areas of stem cell science, machine learning, and computer simulation, as well as new 3D bioprinting hardware and software." It is important to understand that there are still years of research yet to be done, notes Feinberg, "but there should still be excitement that we're making real progress towards engineering functional human tissues and organs, and this paper is one step along that path."
Comments »
If you enjoyed this article, please consider sharing it:
|