3rd September 2019 Scientists regrow tooth enamel Scientists in China report a way of regrowing the complex structure of tooth enamel, using calcium phosphate ion clusters as a precursor layer.
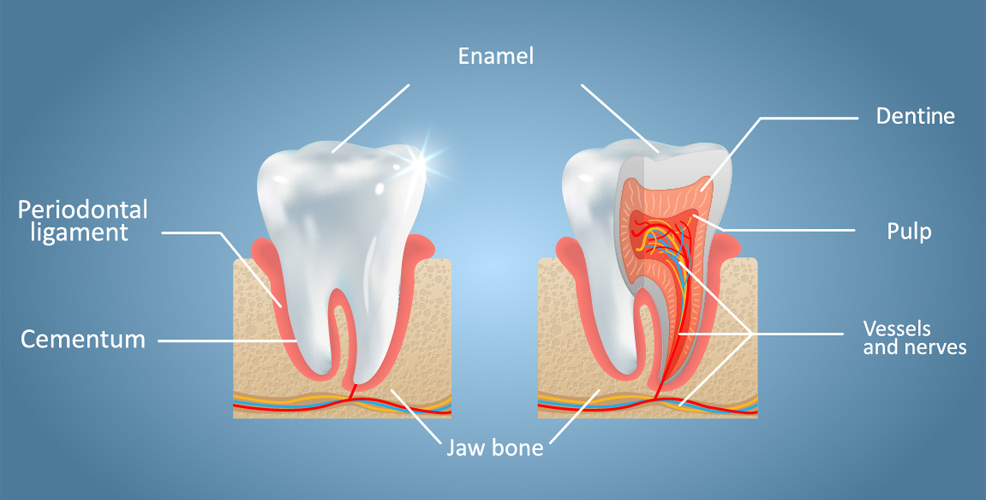
Enamel is the hardest substance in the human body and contains the highest percentage of minerals at 96%. Unlike bones, however, it cannot regenerate and so any damage to its structure is permanent. Cavities are formed when food and drink acids dissolve tooth enamel. Losses also occur from tooth wear and fractures. Regular dental hygiene, by using a toothbrush at least twice daily, is the best way of preventing demineralisation. Flossing between adjacent teeth can also remove plaque and food particles to discourage bacterial growth. The inclusion of fluoride in public water is another effective method. However, only 55% of the world's population owns a toothbrush and just 5% receives artificially-fluoridated water. In any case, neither floss nor toothbrushes can penetrate the deep grooves and pits of enamel. Sugary diets are also on the increase in many countries. This makes tooth decay an extremely common problem: globally, about 2.4 billion people live with caries in permanent teeth, while 486 million children have decay in their milk teeth. Various dental procedures are available to restore the function, integrity and morphology of damaged teeth. These include the use of materials such as amalgam (a liquid mercury and metal alloy mixture), ceramics, gold and resin. Unfortunately, problems can often arise. For example, fillings may become loose just a few years after their placement; a weakening of the overall tooth structure can result in cracked tooth syndrome; and nerves can become overly sensitive to hot and cold substances. These complications, which are often very painful, require further treatment that may be unaffordable to some. For many years, the medical community has been researching ways to reproduce the structure of natural tooth enamel, but its complexity has proved to be a formidable challenge. Now, a team in China has achieved a breakthrough, reported in the journal Science Advances.
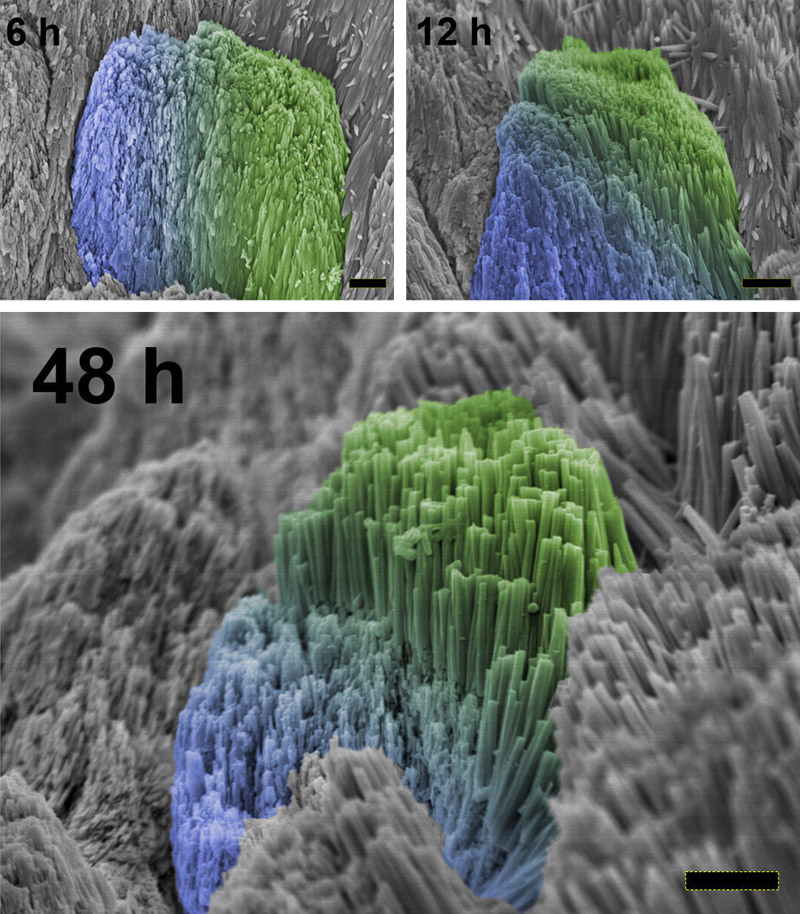
In their study, the researchers describe the creation of extremely tiny clusters of calcium phosphate, the main ingredient of natural enamel. These were successfully produced with diameters of just 1.5-nanometres (nm), far smaller than any previous effort. A chemical compound known as triethylamine was used to prevent the clusters from clumping together. The clusters were combined with a gel, and then tested on crystalline hydroxyapatite, a material very similar to human enamel. Tests showed that the clusters fused with the hydroxyapatite, creating a precursor layer. The structures were confirmed to be much stronger and more tightly organised than in any previous research efforts. They formed a single layer, rather than multiple crystalline areas. The same experiment was later performed using real human teeth, as opposed to a hydroxyapatite sample. These were exposed to acid beforehand to remove their enamel. As seen in the images above, a structure very similar to that of natural enamel began to form. Within 48 hours, the new material had reached a thickness of 2.7 micrometres (μm). Physical testing showed it to be almost identical to natural enamel in strength and wear resistance. This method is cheap and has potential to be scaled up to much greater thicknesses, according to Dr Zhaoming Liu, study co-author from the Zhejiang University in China: "After intensive discussion with dentists, we believe that this new method can be widely used in future," he said. However, the team cautions that more research is needed. Human clinical trials are expected to begin in the next one to two years, but it could be several more years before the first usage in dental surgeries.
Comments »
If you enjoyed this article, please consider sharing it:
|