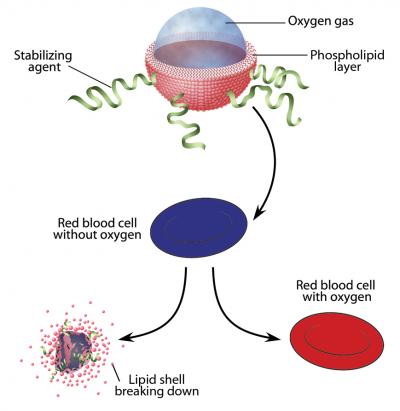
Microparticles allow survival without breathing 28th June 2012 Patients unable to breathe due to lung failure or an obstructed airway need another way to get oxygen to their blood — and fast — to avoid cardiac arrest and brain injury. A team led by researchers at Boston Children's Hospital has designed microparticles that can be injected into the body to quickly oxygenate the blood. These tiny particles consist of a single layer of lipids (fatty molecules), surrounding a pocket of oxygen gas, and are delivered in a liquid solution.
In a cover article published yesterday in Science Translational Medicine, John Kheir and his team report that infusion of these particles into rabbits with low blood oxygen levels restored blood oxygen saturation to near-normal levels, within seconds. When the trachea was completely blocked — a more dangerous "real world" scenario — infusion kept the animals alive for 15 minutes without a single breath, and reduced the incidence of cardiac arrest and organ injury. The particle solutions are portable and could stabilise patients in emergency situations, buying time for paramedics, emergency clinicians or intensive care clinicians to more safely place a breathing tube or perform other life-saving therapies, says Kheir. “This is a short-term oxygen substitute — a way to safely inject oxygen gas to support patients during a critical few minutes,” he says. “Eventually, this could be stored in syringes on every code cart in a hospital, ambulance or transport helicopter, to help stabilise patients who are having difficulty breathing.”
The microparticles would likely only be used for a short time, between 15 and 30 minutes, because they are carried in fluid that would overload the blood if used for longer periods, Kheir says. Kheir also notes that the particles are different from blood substitutes, which carry oxygen but are not useful when the lungs are unable to oxygenate them. Instead, these microparticles are designed for situations where the lungs are completely incapacitated. Kheir began investigating the idea of injectable oxygen in 2006, after caring for a little girl who sustained a major brain injury resulting from pneumonia that caused bleeding into her lungs and severely low oxygen levels. Despite the team’s best efforts, she died before they could place her on a heart-lung machine. Frustrated by this, Kheir formed a team to search for another way to deliver oxygen. “Some of the most convincing experiments were the early ones,” he says. “We drew each other’s blood, mixed it in a test tube with the microparticles and watched blue blood turn immediately red, right before our eyes.”
Over the years, Kheir and his team have tested various concentrations and sizes of the microparticles to optimise their effectiveness and make them safe for injection. “The effort was truly multidisciplinary,” says Kheir. “It took chemical engineers, particle scientists and medical doctors to get the mix just right.” In the studies reported in the paper, they used a device called a sonicator, which uses high-intensity sound waves to mix the oxygen and lipids together. This process traps oxygen gas inside particles 2 to 4 micrometers in size (not visible without a microscope). The resulting solution, with oxygen gas making up 70% of the volume, mixed efficiently with human blood. “One of the keys to the success of the project was the ability to administer a concentrated amount of oxygen gas in a small amount of liquid,” Kheir says. “The suspension carries three to four times the oxygen content of our own red blood cells.” Intravenous administration of oxygen gas was tried in the past, but these attempts failed to oxygenate the blood and often caused dangerous gas embolisms. “We have engineered around this problem by packaging the gas into small, deformable particles,” Kheir explains. “They dramatically increase the surface area for gas exchange and are able to squeeze through capillaries where free gas would get stuck.” The capsules are cheap and easy to make, he says. They can self-assemble during the sonication process. If this method works in larger animals and is successful in human trials, it could eventually save millions of lives each year.
Comments »
|