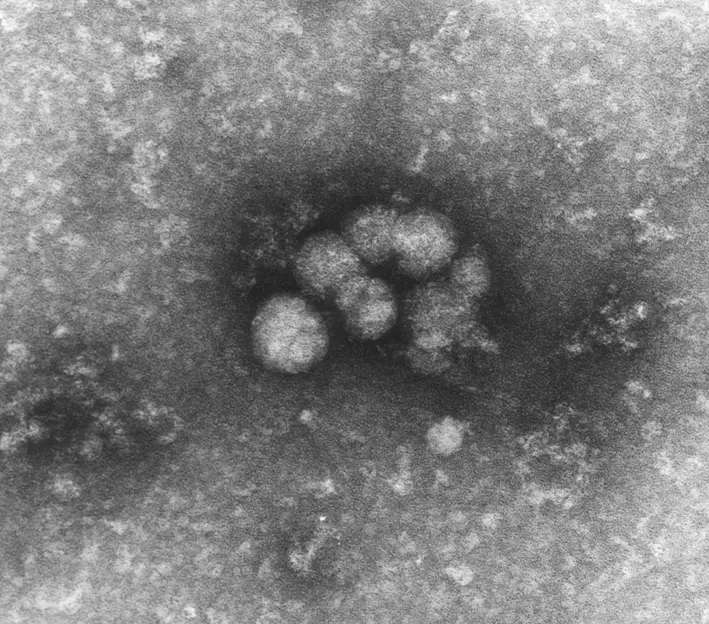
19th May 2013 New treatments for hepatitis C Several emerging treatments for this debilitating illness could dramatically improve cure rates, while greatly decreasing the side effects and period of time needed to recover. It is estimated that between 130–200 million people – or nearly 3% of the world's population – are living with chronic hepatitis C. The disease was first postulated in the 1970s and proven in 1989. The hepatitis C virus (HCV) is spread primarily by blood-to-blood contact from intravenous drug use, poorly sterilised medical equipment and transfusions. Affecting mainly the liver, it is often asymptomatic, but can lead to eventual scarring of the liver and ultimately to cirrhosis, liver failure, cancer and other life-threatening conditions. No vaccine against hepatitis C is available. HCV induces chronic infection in 50–80% of cases. Of these, around 75% will clear with medication. However, lengthy periods of treatment are often required – up to 48 weeks or more, depending on the genotype. Adverse side effects are common, with half of people getting flu-like symptoms and a third experiencing emotional problems. The economic costs are significant both to the individual and society. In the USA for example the average lifetime cost of the disease has been estimated at $33,407, with a liver transplant costing $200,000. At the Conference on Retroviruses and Opportunistic Infections (CROI), held recently in Atlanta, a range of new studies on direct-acting antivirals (DAAs) was highlighted. They are summarised in the abstract of a paper which states:
Interferon is an immune-boosting drug used in current treatments – and is responsible for many of the side effects described earlier – so its replacement with new, direct-acting drugs like those mentioned here would be a major boost in terms of curing HCV.
Comments »
|