3rd April 2021 Artificial life can grow and divide normally A breakthrough in synthetic biology could shed new light on mechanisms controlling the most basic processes of life.
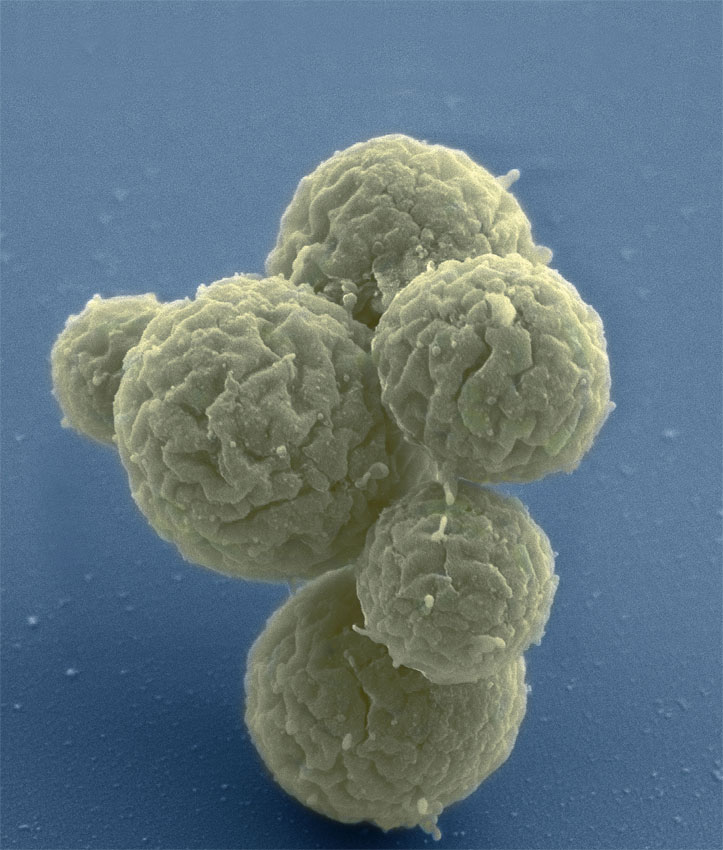
In March 2016, scientists created a new, synthetic, single-celled organism. With only 473 genes, this became the first "minimal" synthetic bacterial cell and the simplest living cell ever known. For comparison, a human cell has over 20,000 genes. However, while undoubtedly a landmark scientific breakthrough, this bacteria-like organism behaved strangely when growing and dividing – producing cells with wildly different shapes and sizes. Now, scientists have identified seven genes that can be added to make the cells organise themselves better and divide neatly, into uniform orbs. This new discovery is the result of a collaboration between the J. Craig Venter Institute (JCVI), the National Institute of Standards and Technology (NIST) and the Massachusetts Institute of Technology (MIT) Center for Bits and Atoms. The researchers have described their work in the journal Cell. Identifying these genes is an important step towards engineering synthetic cells that do useful things. Such cells could act as small factories that produce drugs, foods and fuels; detect disease and produce drugs to treat it while living inside the body; or even function as tiny computers.
To design and build a cell that does exactly what you want it to do, it helps to have a list of essential parts and know how they fit together. "We want to understand the fundamental design rules of life," said Elizabeth Strychalski, co-author and leader of NIST's Cellular Engineering Group. "If this cell can help us to discover and understand those rules, then we're off to the races." Scientists at JCVI constructed the first cell with a synthetic genome in 2010. They started with cells from a very simple type of bacteria called a mycoplasma and then destroyed the DNA in those cells, before replacing it with DNA designed on a computer and synthesised in a lab. This became the first organism in the history of life on Earth to have an entirely synthetic genome and was named JCVI-syn1.0. Since then, scientists have been working to strip that organism down to its minimum genetic components. The super-simple cell they created five years ago – dubbed JCVI-syn3.0 – was perhaps too minimalist. In their latest work, the researchers have added 19 genes back to this cell, including the seven needed for normal cell division. This variant, known as JCVI-syn3A, now has 492 genes. To put that number in perspective, the E. coli bacteria that live in your gut have about 4,000 genes. Identifying those seven additional genes took years of painstaking effort by JCVI's synthetic biology group. The team constructed dozens of variant strains by systematically adding and removing genes, then observed how those genetic changes affected cell growth and division. Living cells are difficult to image over long periods, because they are so small and delicate. To keep these cells alive and held in place, the scientists designed a microfluidic chemostat – a sort of mini-aquarium – where the cells could be kept fed and happy under a light microscope.
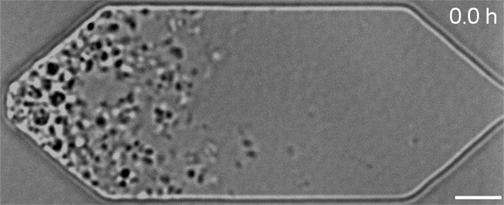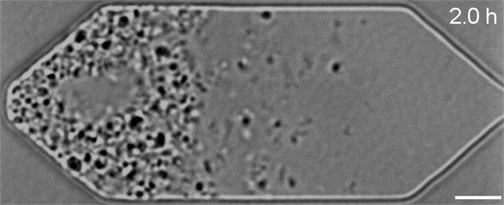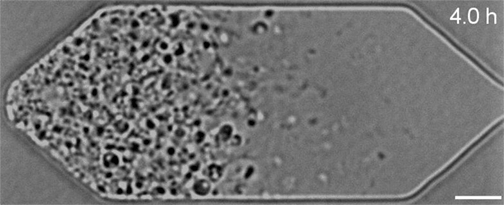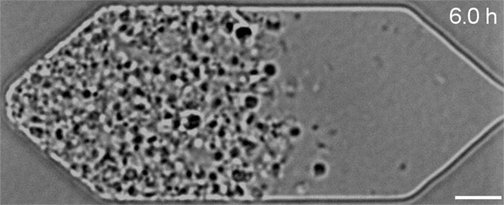
In the video below, all three synthetic organisms are compared. The original JCVI-syn1.0 (created in 2010) is shown first. The minimal version JCVI-syn3.0 (created in 2016) is next. Finally, the new version JCVI-syn3A with 19 additional genes is shown last. As can be seen, the minimal version 3.0 from 2016 divides into many different shapes and sizes. Some of the cells form filaments. Others appear to not fully separate and line up like beads on a string. Despite the variety, all these cells are genetically identical. The new version 3A, however, clearly divides into cells of more uniform shape and size. It looks and divides like the wild type and JCVI-syn1.0. "Designing whole genomes to achieve desired phenotypes stands as a grand challenge in synthetic biology," said Dr. Strychalski. "But our capacity to synthesise and modify genomes has rapidly outpaced our ability to predict phenotype from genotype for large-scale genome design. Our work uses reverse genetics to understand the function of genes of unknown function involved in the basic cell processes of controlling cell size and shape, and cell division. Every gene we are able to pair with its function gets us closer to realising the goal of designing genomes for engineering cells." JCVI-syn3.0 and JCVI-syn3A now provide a robust platform for investigating how modern cell division and cell size evolved. Both the minimal cell and the new synthetic cell have been distributed for use in research at more than 40 labs worldwide. This includes adapted laboratory evolution experiments and work on membrane composition, as well as whole cell computational modelling. In addition, the cells are already being explored for commercial application.
Comments »
If you enjoyed this article, please consider sharing it:
|