1st June 2013 The first-ever high-resolution images of a molecule as it breaks and reforms chemical bonds Every chemist’s dream – to capture an atomic-scale picture of a chemical before and after it reacts – has now come true, thanks to a new technique developed by researchers at the University of California.
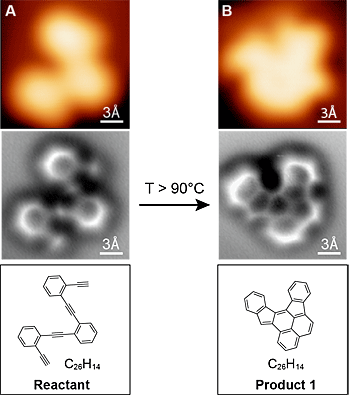
Using a state-of-the-art, atomic force microscope, the scientists have taken the first atom-by-atom pictures, including images of the chemical bonds between atoms, clearly depicting how a molecule's structure changed during a reaction. Until now, scientists have only been able to infer this type of information from spectroscopic analysis. “Even though I use these molecules on a day to day basis, actually being able to see these pictures blew me away. Wow!” said lead researcher Felix Fischer, UC Berkeley assistant professor of chemistry. “This was what my teachers used to say that you would never be able to actually see, and now we have it here.” The ability to image molecular reactions in this way could help not only students as they study chemical structures and their reactions, but will also show chemists the products of their experiments and help them fine-tune the reactions to get the results they want. Fischer, along with Professor Michael Crommie, captured these images with the goal of building new graphene nanostructures – a hot area of research today for their potential in next-generation computers. However, the implications go far beyond just graphene, Fischer said. This technique will find application in the study of heterogeneous catalysis, for example, which is used widely in the oil and chemical industries. Heterogeneous catalysis involves the use of metal catalysts like platinum to speed up reactions, as in the catalytic converter of a car. “To understand the chemistry that is actually happening on a catalytic surface, we need a tool that is very selective and tells us which bonds have actually formed and which ones have been broken,” Fischer explained. “This technique is unique out there right now for the accuracy with which it gives structural information. I think it’s ground-breaking.”
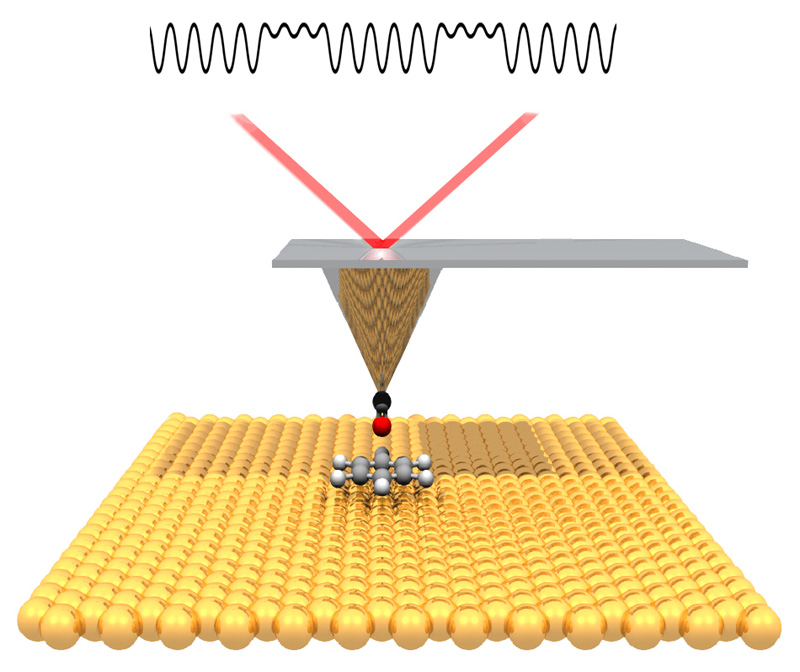
“The atomic force microscope gives us new information about the chemical bond, which is incredibly useful for understanding how different molecular structures connect up, and how to convert from one shape into another shape,” said Crommie. “This should help us to create new engineered nanostructures – such as bonded networks of atoms that have a particular shape and structure for use in electronic devices. This points the way forward.” Traditionally, researchers conduct detailed analyses to determine the products of a chemical reaction – and even then, the actual 3-D arrangement of atoms in these products can be ambiguous. “In chemistry, you throw stuff into a flask and something else comes out, but you typically only get very indirect information about what you have,” Fischer said. “You have to deduce that by taking nuclear magnetic resonance, infrared or ultraviolet spectra. It is more like a puzzle, putting all the information together and then nailing down what the structure likely is. But it's just a shadow. Here, we actually have a technique at hand where we can look at it and say this is exactly the molecule. It’s like taking a snapshot of it.” Working together, Fischer and Crommie devised a way to chill the reaction surface and molecules to the temperature of liquid helium, which stops them from jiggling around. They then used a scanning tunnelling microscope to locate all the molecules on the surface, and zeroed in on several to probe more finely with the atomic force microscope. To enhance the spatial resolution, they put a single carbon monoxide molecule on the tip. After imaging the molecule, they heated the surface until the molecule reacted, then again chilled the surface and imaged the reaction products. The study was published this week in Science.
Comments »
|