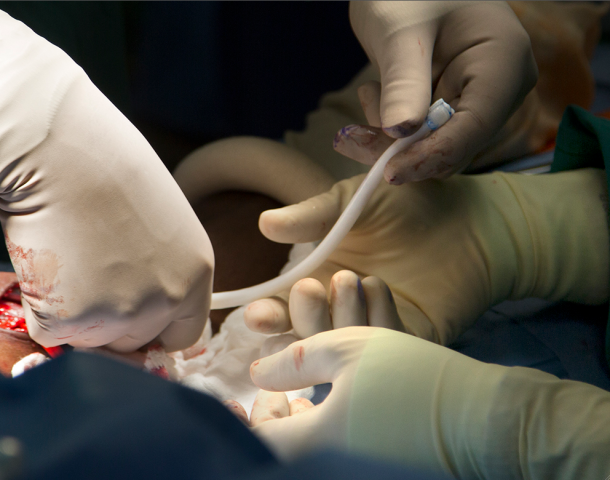
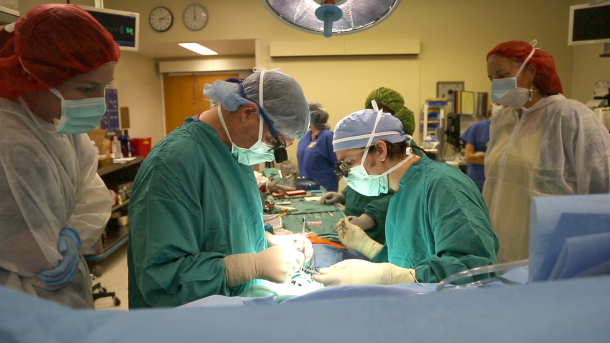
7th June 2013 Bioengineered vein implant is a milestone in tissue engineering A kidney dialysis patient has become the first in the U.S. to receive a lab-grown blood vessel. It is hoped this could be a possible stepping stone toward more complex organs such as livers or eyes.
In a first-of-its-kind operation in the United States, a team of doctors at Duke University Hospital have created a bioengineered blood vessel which was transplanted into the arm of a patient with end-stage kidney disease. The procedure – the first U.S. clinical trial to test its safety and effectiveness – is a milestone in the field of tissue engineering. The new vein is an off-the-shelf, human cell-based product with no biological properties that would cause organ rejection. Using technology developed in a 15-year collaboration between Duke and a spin-off company it started called Humacyte, the vein is engineered by cultivating donated human cells on a tubular "scaffold" to form a vessel. This vessel is then cleansed of the qualities that might trigger an immune response. In pre-clinical tests, the veins have performed better than other synthetic and animal-based implants. "This is a pioneering event in medicine," said Jeffrey H. Lawson, M.D., PhD, a vascular surgeon and vascular biologist at Duke Medicine who helped develop the technology and performed the implantation. "It's exciting to see something you've worked on for so long become a reality." Clinical trials to test the new veins began in Poland in December with the first human implantations. The U.S. Food and Drug Administration recently approved a phase 1 trial involving 20 kidney dialysis patients in the United States, followed by a safety review. Duke researchers enrolled the first U.S. patient and serve as study leaders. The initial trial focuses on implanting the vessels in an easily accessible site in the arms of kidney hemodialysis patients. Over 320,000 people in the U.S. require hemodialysis, which often necessitates a graft to connect an artery to a vein to speed blood flow during treatments. If the bioengineered veins prove beneficial for hemodialysis patients, the researchers ultimately aim to develop a readily available and durable graft for heart bypass surgeries, which are performed on nearly 400,000 people in the U.S. a year, and to treat blocked blood vessels in the limbs. “We hope this sets the groundwork for how these things can be grown, how they can incorporate into the host, and how they can avoid being rejected immunologically,” said Lawson. “A blood vessel is really an organ – it’s complex tissue. We start with this, and one day we may be able to engineer a liver or a kidney or an eye.” More complex bioengineered organs have already been tested on rodents. In April, for example, scientists at the Massachusetts General Hospital in Boston grew a kidney that was transplanted back into a rat, where it began producing urine. Experts believe this could start to become possible in humans by 2020.
Comments »
|