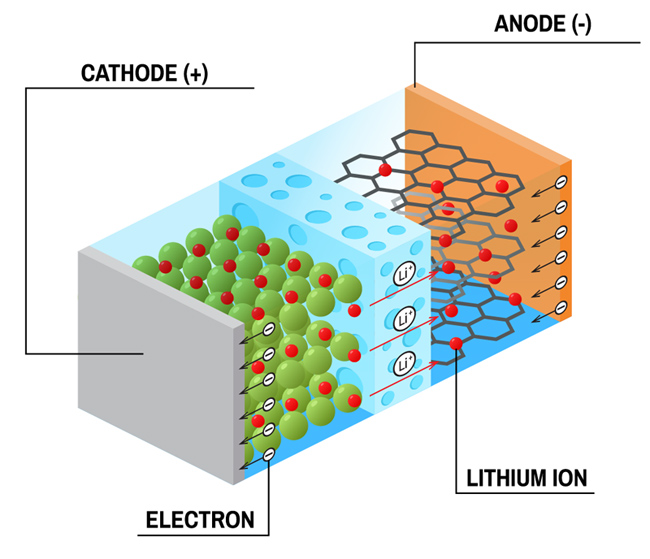
17th July 2020 New cobalt-free lithium-ion battery reduces costs without sacrificing performance Researchers at the University of Texas at Austin have demonstrated a cobalt-free, high-energy, lithium-ion battery.
For decades, researchers have looked for ways to eliminate cobalt from the high-energy batteries that power electronic devices, due to its high cost and the human rights ramifications of its mining. But past attempts have been unable to reach the performance standards of batteries with cobalt. This month, however, the Cockrell School of Engineering at the University of Texas at Austin (UTA) reports a potentially major breakthrough. A team of researchers believe they have cracked the code to a cobalt-free, high-energy, lithium-ion battery – eliminating the need for cobalt and opening the door to reducing the costs of producing batteries, while actually boosting performance in some ways. Key to this development is a new class of cathodes (the electrode in a battery where cobalt typically resides), anchored by high nickel content. The cathode in their study is 89% nickel, with manganese and aluminium making up the other key elements. More nickel in a battery means it can store more energy. That increased energy density could lead to longer battery life for a phone, or greater range for an electric vehicle with each charge.
Typically, increased energy density leads to trade-offs, such as a shorter cycle life – the number of times a battery can be charged and discharged before it loses efficiency and is no longer fully chargeable. Eliminating cobalt usually slows down the kinetic response of a battery, leading to lower rate capability – how quickly the cathode can be charged or discharged. However, the researchers say they have overcome the short cycle life and poor rate capability problems by finding an optimal combination of metals and ensuring an even distribution of their ions. Most cathodes for lithium-ion batteries use combinations of metal ions, such as nickel-manganese-cobalt (NMC) or nickel-cobalt-aluminium (NCA). Cathodes can make up roughly half of the material costs for the entire battery, with cobalt being the priciest element. At a price of approximately $28,500 per ton, it is more expensive than nickel, manganese and aluminium combined, and it makes up 10% to 30% of most lithium-ion battery cathodes. "Cobalt is the least abundant and most expensive component in battery cathodes," explains Arumugam Manthiram, a professor in the Walker Department of Mechanical Engineering at UTA and director of the Texas Materials Institute. "And we are completely eliminating it." The researchers achieved their breakthrough by working at the atomic level. During synthesis, they were able to ensure the ions of the various metals remained evenly distributed across the crystal structure in the cathode. When these ions bunch up, performance degrades, a problem that has plagued earlier cobalt-free, high-energy batteries. By keeping the ions evenly distributed, the researchers avoided performance loss.
"Our goal is to use only abundant and affordable metals to replace cobalt, while maintaining the performance and safety," said PhD graduate, Wangda Li, "and to leverage industrial synthesis processes that are immediately scalable." The team has now formed a startup called TexPower to bring the technology to market. The researchers have received grants from the US Department of Energy, which has sought to decrease dependency on imports for key battery materials. Industry has jumped on the cobalt-free push in recent years – notably Tesla, which is aiming to eliminate the material from the batteries that power its electric vehicles. With large government organisations and private companies increasingly focused on reducing dependence on cobalt, it should be unsurprising that this pursuit has become competitive. The team at UTA have solved the problems that hindered previous attempts, using the right combination of materials and precise control of their distribution. This milestone could help in meeting the exponentially growing demand for batteries, which are set to become a key technology of the 2020s and 2030s. "We are increasing the energy density and lowering the cost, without sacrificing cycle life," adds Professor Manthiram. "This means longer driving distances for electric vehicles and better battery life for laptops and cellphones."
Comments »
If you enjoyed this article, please consider sharing it:
|