25th February 2026 First benchtop $100 genome system The long-promised $100 genome may finally be entering mainstream commercial territory. California-based Element Biosciences has launched what it describes as the first benchtop sequencing platform explicitly designed to deliver whole human genomes at that price – a step beyond earlier room-scale demonstrations by rival firms.
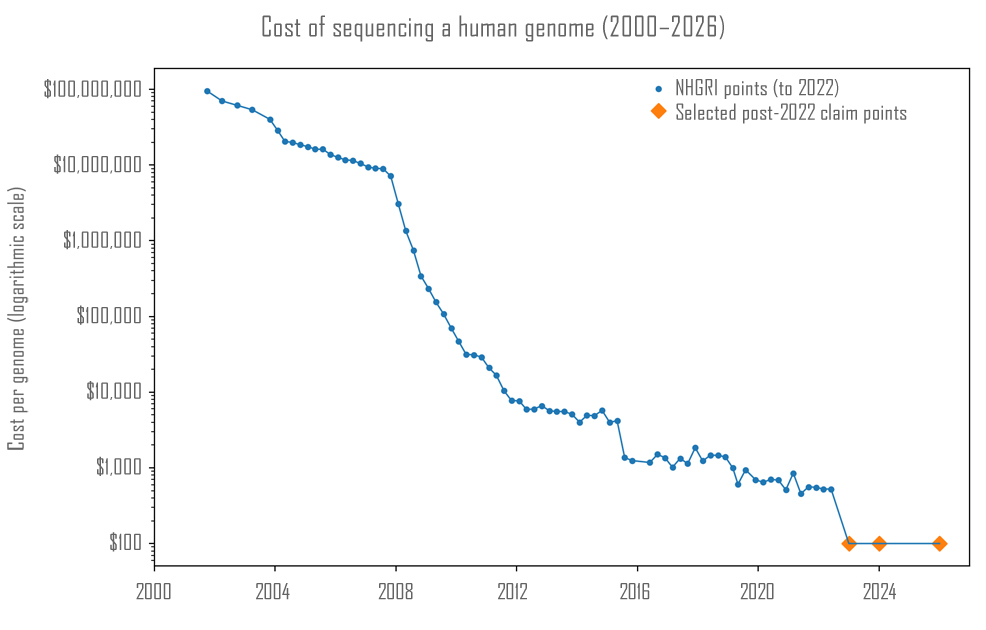
In 2003, scientists involved in the Human Genome Project completed the first full human genome sequence. The effort had taken 13 years of global collaboration and required $2.7 billion in funding. It provided the first complete reference map of human DNA and laid the foundation for modern genomics. What followed ranks among the steepest technological cost declines in modern history. Between 2007 and 2012, the price of sequencing a human genome plunged at a rate that far outpaced Moore's Law – declining by several orders of magnitude to just a few thousand dollars – as companies improved sequencing chemistry, increased parallelisation and refined high-throughput platforms. By the mid-2010s, however, the downward curve began to flatten. Providers typically charged between $600–$1,000 for high-quality whole-genome sequencing at 30× coverage, meaning each part of the genome was read about thirty times to ensure accuracy. In recent years, the decline has resumed. At least two companies have demonstrated that laboratories can approach $100-level genome economics, but only under very high-throughput research conditions using bulky, room-scale machines. This month, California-based Element Biosciences, founded in 2017, has introduced VITARI, describing it as "the first ever high-throughput benchtop sequencing system capable of delivering a high-quality, whole human genome at $100." The compact platform now brings large-scale sequencing capacity directly into individual laboratories rather than concentrating it in vast centralised facilities.
"VITARI is about removing limits from scientific ambition," said Molly He, CEO and co-founder. "By lowering the barriers to high-throughput sequencing for labs of all sizes, we're improving workflows and giving researchers the freedom to design studies around biology – not the constraints of their tools. And with an architecture built to expand functionality, VITARI will grow with the applications scientists need next." Element has engineered VITARI to deliver up to 10 billion DNA reads per dual-sided run, generating as much as 3 terabytes of data. Each read represents a short fragment of DNA sequence, and software assembles billions of these fragments into a complete genome. At this level of throughput, the device can sequence multiple human genomes at standard 30× coverage in a single run. Element has set the machine's price at $689,000 and plans to begin shipping in the second half of 2026, with pre-orders already open. VITARI's exact dimensions and weight have yet to be published by Element Biosciences. As a compact benchtop platform, however, it represents a stark contrast with previous "$100 genome" efforts. Complete Genomics, for example, developed an industrial system called the DNBSEQ-T20×2, spanning several metres and weighing around 3.7 tonnes. Rival company Ultima Genomics' UG 100 setup relies on floor-standing modules, with key components weighing hundreds of kilograms each. By contrast, VITARI is the first $100 genome system to fit on a laboratory benchtop. This transition echoes the computer revolution of the 1970s, when room-sized mainframes shrank to desktop form and broadened access to computing power. The implications extend beyond form factor and cost. In the near term, improved accessibility of whole-genome sequencing could expand population-scale research programmes, rare disease studies, and cancer genomics. Hospitals and regional laboratories that previously relied on centralised sequencing hubs may gain greater autonomy, shortening turnaround times and lowering logistical barriers. Looking further into the future, continued cost reductions could enable the integration of genomics into everyday healthcare. If whole-genome sequencing falls well below the $100 threshold and analysis tools continue to improve, clinicians may one day treat an individual's genome as a standard medical reference, consulted throughout life for disease risk, drug response and early detection strategies. Just as computing power moved from national laboratories to desktops and eventually into pockets, the reading of human DNA may follow a similar trajectory – shifting from specialised facilities to routine infrastructure underpinning preventive and personalised medicine. More than twenty years after the completion of the Human Genome Project, sequencing technology continues to defy expectations. The $100 genome now appears within reach of ordinary laboratories, and further cost reductions could push the field toward $10 or even $1 thresholds in the decades ahead. What once required billions of dollars and a consortium of the world's leading experts may become commonplace in modern medicine.
Comments »
If you enjoyed this article, please consider sharing it:
|
||||||