25th March 2026 World's first lab-grown oesophagus In recent years, scientists have made steady progress towards growing replacement body parts in the lab, but complex tubular organs have remained especially difficult. Now, a team in London has taken a major step forward by demonstrating the first lab-grown oesophagus able to restore swallowing in a large animal.
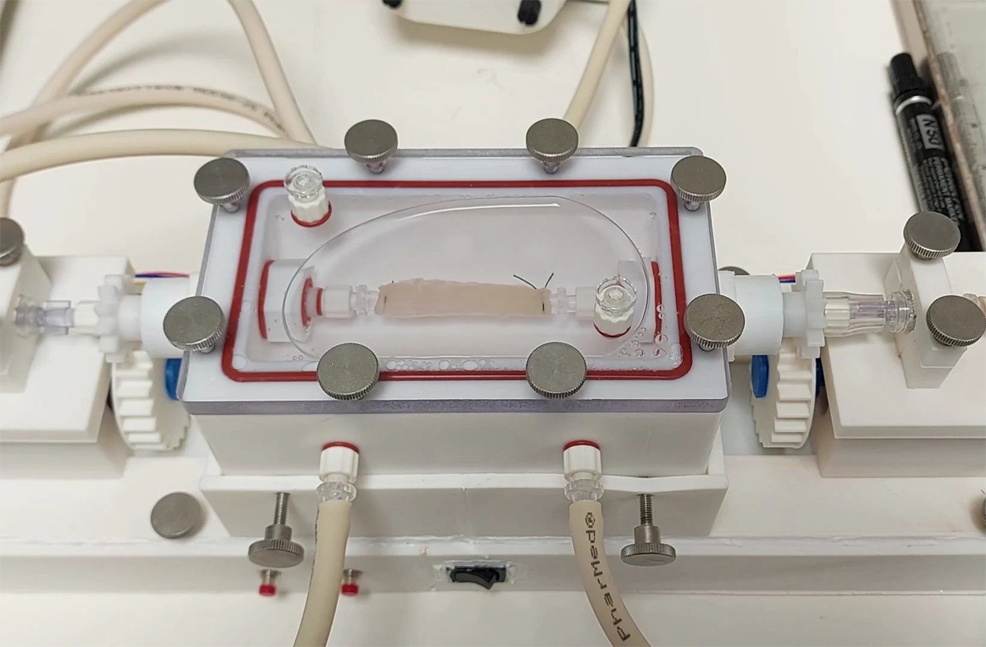
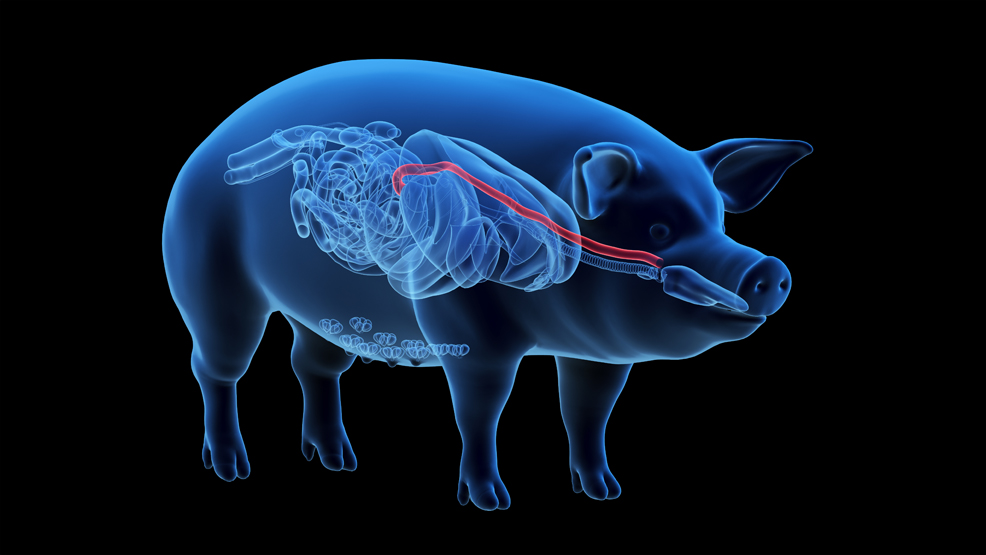
For decades, scientists have worked towards building tissues that can repair or replace damaged parts of the body. They have made considerable progress with relatively simple tissues, but the oesophagus presents a much tougher challenge. It is not just a hollow tube. It must survive constant movement, carry food to the stomach, develop muscle and nerve function, and integrate with surrounding tissue during growth. That is why a new study from Great Ormond Street Hospital (GOSH) and University College London (UCL), published in Nature Biotechnology, stands out. Researchers created a lab-grown oesophagus and implanted it into pigs, where it successfully restored function without the need for immunosuppressive drugs. While the experiments took place in animals, the work aims to develop a new treatment for children born with long-gap oesophageal atresia (LGOA), a rare and life-threatening condition in which part of the food pipe is missing. Current treatments remain difficult and invasive, often requiring major surgery to reposition the stomach or intestine. These procedures can save lives, but they may also bring long-term complications affecting breathing, digestion and quality of life. The new approach offers a more tailored alternative. First, the team took an oesophagus from a donor pig and removed all its cells, leaving behind only the structural scaffold. This process, known as decellularisation, preserves the organ's natural architecture while removing the biological material most likely to trigger immune rejection.
The researchers then seeded the scaffold with muscle cells taken from the recipient animal, grew them in the lab, and placed the construct in a bioreactor that pumped nutrients through the tissue for a week. In total, the process took around two months. The greatest challenge was not simply creating a replacement tube, but building one that could become living, functional tissue. In all eight implanted animals, that appears to have happened. By six months, the grafts had developed muscle, nerves, and blood vessels. They contracted with enough strength and coordination to move food normally, and the animals were able to eat and grow healthily. While some developed narrowing (strictures), these were successfully managed through endoscopy, mirroring routine human clinical practice. The researchers also used spatial transcriptomics to examine gene activity within the grafts, confirming that the tissue had matured in a way consistent with a natural oesophagus. This adds an important layer of evidence that the team achieved true regeneration, not merely temporary repair. The team still needs to complete several steps before this reaches patients. They now aim to refine manufacturing, improve blood flow, produce longer grafts and carry out further safety testing before human trials. Even so, this represents one of the clearest signs yet that personalised, lab-grown organs may one day become a practical reality. For children facing repeated operations and years of complications, that future cannot come soon enough. Dr Marco Pellegrini, Senior Researcher at UCL and study co-leader, said: "Our technology could allow us to build a child a new oesophagus, using their own cells, collected in a surgery they are having anyway, combined with a ready-prepared scaffold from pig tissue. Because the graft contains the child's own muscle progenitor cells, it would be recognised as their own tissue. This means it could grow with them over time, without the risk of rejection and without the need for long-term immunosuppression." Prof Paolo De Coppi, Head of Regenerative Medicine at the GOSH Biomedical Research Centre, said: "For more than 50 years, pig heart valves have been used to extend and save the lives of patients with heart disease, and this technology is now commonplace in cardiac surgery. More recently, xenotransplantation has been explored in humans as a potential solution to organ shortages. In our work, we demonstrate that pig tissue, once stripped of all cellular material, can serve as a scaffold to engineer humanised tissue that is fully biocompatible. I believe we are now standing at a similar new frontier in regenerative medicine. With the success of this research, we hope that we can be offering an engineered tissue alternative to children who desperately need it, within five years."
Comments »
If you enjoyed this article, please consider sharing it:
|
||||||